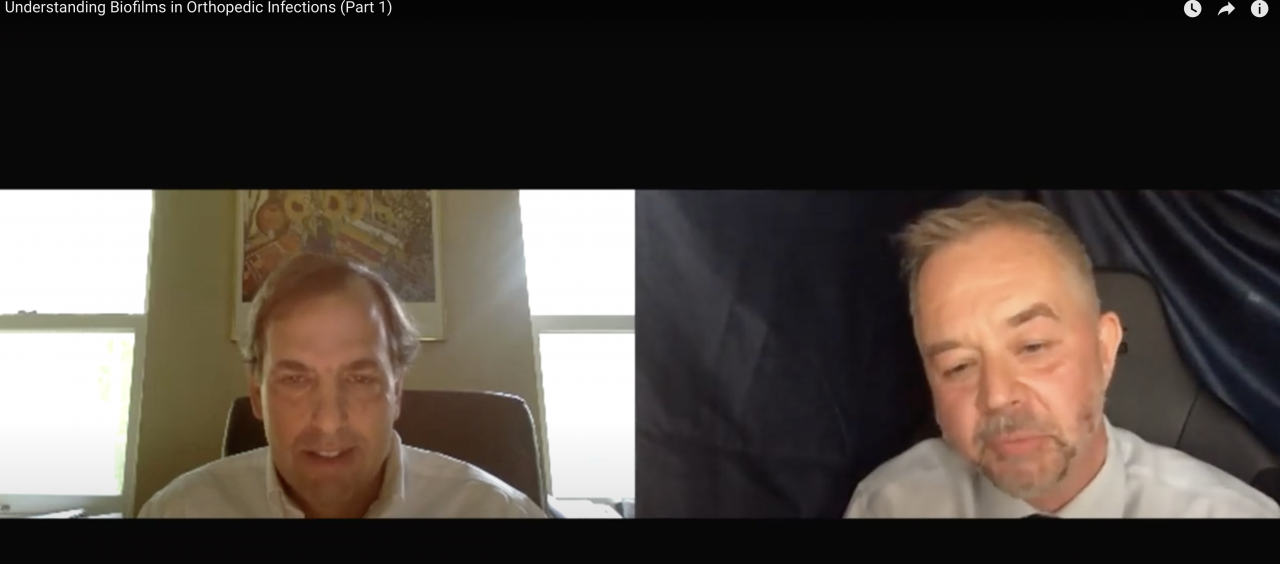
We provide world leading microbiological consultancy and bespoke contract research services, by delivering efficient and effective Biofilm, Antimicrobial, Wound Care, MedTech and Virology research.
Our partnership approach supports our clients’ to enable them to commercialise and develop innovative, life-changing new products to help protect the health of our population from unnecessary infections.